A new method identifies active agents in mixtures of hundreds of substances
Poison for cancer cells
In their quest for new agents, pharmaceutical researchers test millions of substances all over the world. They like using color-forming reactions to identify new molecules. However, in intensively colored solutions or in the case of mixtures with multiple substances these tests fail. As part of his doctoral thesis, Martin Stein, member of staff at the Chair of Biochemistry at the Technische Universität München, developed a testing reaction based on magnetic resonance data. It helps find a specific pharmaceutical molecule among hundreds of different substances even in the most turbid of bacterial brews.
As a test reaction, the research team used the decomposition of a short amino acid chain caused by the proteasome. Similar to a recycling facility this vitally important cell protein breaks down superfluous proteins into small pieces. To follow the reaction, they integrated a special carbon atom into the chain: it has one neuron more than normal carbon and its magnetic resonance signal is sensitive to a scission of the chain. Under normal circumstances, the proteasome will decompose the amino acid chain, effecting a change in the signal from the carbon-13 atom. If it does not react, the solution must contain a substance that inhibits the work of the proteasome.
Cancer cells suffocating in garbage
The pharmaceutical industry is always on the lookout for precisely such substances to deploy them against threads like cancer. In the case of cancer, for example, when the proteasome is blocked, rapidly growing cancer cells choke on their own waste. The first medication of this kind is already generating annual revenues of over one billion US dollars. The scientists are now looking for further substances with lesser side effects.
Following preliminary studies, one such candidate was a toxic substance produced by the bacterium Photorhabdus luminescens. This is the poison that kills the larvae of the garden chafer. Using his new methodology, the scientists discovered that the bacterium lives inactively in the intestines of the threadworm. When it lays its eggs, the worm infects the larvae. The sudden change in environment causes the bacterium to emit toxins. After the larva dies, the bacterium ceases to produce toxins. Once the threadworms hatch from the protective egg membrane, they ingest the inactive bacterium into their intestines, and the cycle can start again.
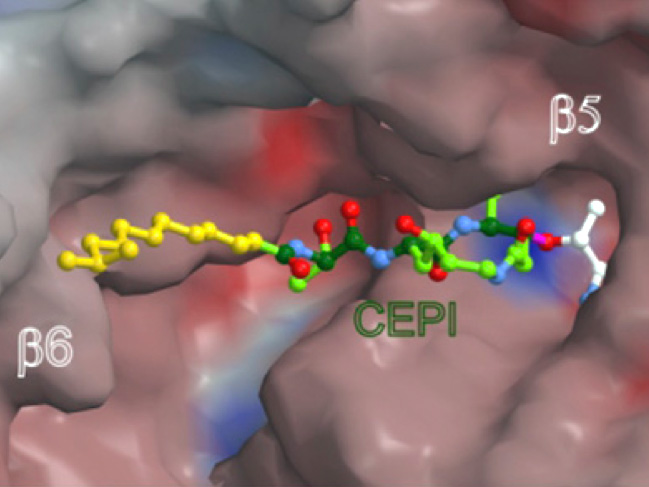
Since the newly developed method also works in intensively colored solutions and in the presence of hundreds of other substances, the workgroup at the Chair of Biochemistry succeeded in isolating the unknown poison directly from the bacterial brew: It turned out to be two structurally very similar compounds, cepafungin I and glidobactin A. The latter was previously considered the strongest proteasome blocker. In spite of the resemblance, cepafungin I had never been tested as a proteasome blocking agent. The tests of the research group showed that Cepafungin I is indeed a strong Proteasomhemmer. In effect, it even surpasses the previous record holder.
Production by bacteria
The production of elaborate pharmaceutical substances via bacteria is the silver bullet in pharmaceutical production. However, there is one catch: Just like with the bacterium Photorhabdus luminescens, the sought-after natural substances are produced in significant amounts during only one phase of the bacterial life cycle. But here, too, the new method has a decisive advantage: “A measurement cycle takes approximately 15 minutes, which means almost 100 samples can be analyzed per day,” says Martin Stein. “This allows us to determine very quickly the circumstances under which the bacterium will yield an optimal amount of the active agent”.
The bacterium Pseudomonas syringae also counts on the deadly effect of its proteasome inhibitor. When the bacterium attacks a bean plant, its leaves turn brown and die. This is caused by the substance syringolin A, which has a basic structure very similar to that of cepafungin I and glidobactin A. It inhibits the proteasome of the plant cell and thus blocks the plant’s defense against the brown spot disease.
“We gained an important insight from these tests,” says Prof. Michael Groll, head of the research team. “Nature has already optimized the basic structure of these substances to inhibit the proteasome. It works in plants as well as in insects and even in people. If we center our research into new agents on these natural substances, we could save valuable time in medication development.”
The work was funded by the Cluster of Excellence Center for Integrated Protein Science Munich (CIPSM). The crystal structures of proteasome complexes were determined using the Swiss Light Source at the Paul Scherrer Institute (Villigen, Switzerland).
Original publication:
One-shot NMR analysis of microbial secretions identifies highly potent proteasome inhibitor; Martin L. Stein, Philipp Beck, Markus Kaiser, Robert Dudler, Christian F. W. Becker and Michael Groll, PNAS, Vol. 109, 45, 18367-71
DOI: 10.1073/pnas.1211423109
Contact:
Prof. Dr. Michael Groll
Technische Universität München
Chair of Biochemistry
Lichtenbergstr. 4
85748 Garching, Germany
Tel.: +49 89 289 13361
Fax: +49 89 289 13363
E-Mail - Internet