Cause of early cellular dysfunction in Alzheimer’s disease recognized for the first time
Direct toxic action of beta-amyloid identified

The brains of Alzheimer's patients who have already developed clinical symptoms contain large clumps of the protein beta-amyloid, known as plaques. Many therapeutic approaches focus on removing plaques, but such attempts have met with only limited success to date.
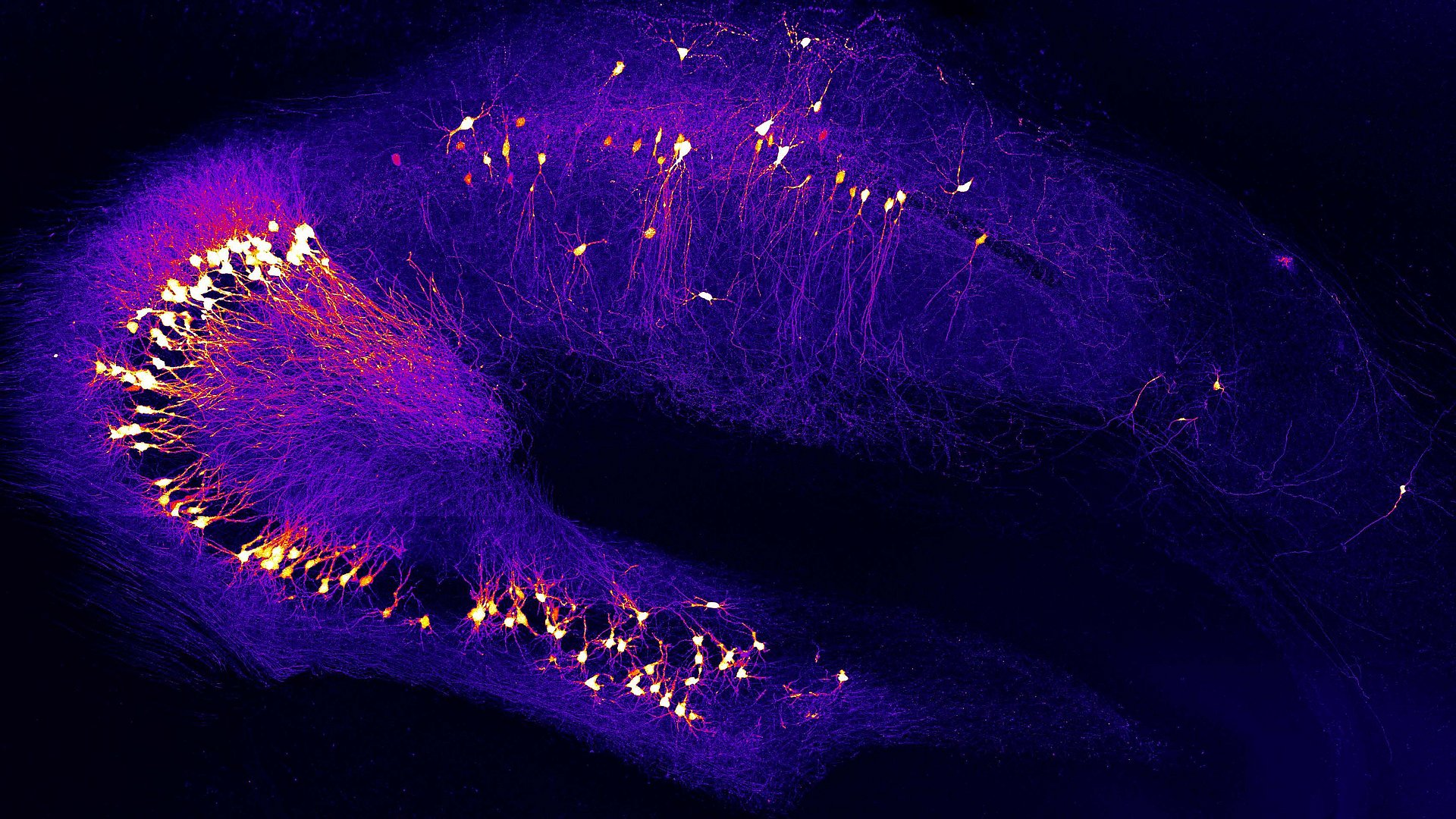
“It’s crucial that we detect and treat the disease much earlier. We therefore focused on hyperactive neurons, which occur at a very early stage – long before patients develop memory loss,” explains Professor Arthur Konnerth, Hertie Senior Professor of Neuroscience at the TUM. As a consequence of hyperactivation, connected neurons in the circuits constantly receive false signals, leading to impairments in signal processing.
Together with his doctoral student Benedikt Zott and the entire research team, Konnerth succeeded in identifying the cause and trigger of this early disturbance in the brain. The discovery may open the way to new therapeutic approaches. The study appeared in the journal Science.
Beta-amyloid blocks glutamate re-uptake
Neurons use chemicals called neurotransmitters to communicate with each other. Glutamate, one of the most important of these chemicals, serves to activate connected neurons. Glutamate is released at the connecting site between two neurons, called synapse, and than rapidly removed to allow the transmission of the next signal. This removal involves so-called active pump molecules as well as a passive transport of glutamate along nearby membranes.
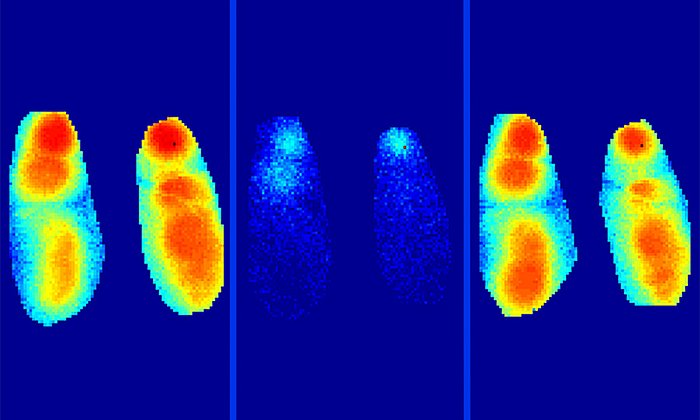
The researchers discovered that high concentrations of glutamate persisted too long in the synaptic cleft of hyperactive neurons. This was due to the action of beta-amyloid molecules, which blocked glutamate transport out of the synaptic cleft. The team tested the mechanism using beta-amyloid molecules from patient samples and by using various mouse models obtaining similar results with both approaches.
Indication for treatment strategies at early stages
The team was also intrigued to discover that the neurotransmitter blockade was mediated by an early soluble form of beta-amyloid and not by the plaques. Beta-amyloid occurs initially as a single molecule form, or monomer, and then aggregates to double-molecule forms (dimers) and larger chains resulting, eventually, in plaques. The researchers found that glutamate blockade is caused by the soluble dimers.
"Our data provide clear evidence for a rapid and direct toxic effect of a particular beta-amyloid type, the dimers. We were even able to explain this mechanism,” as Benedict Zott, first author of the study, outlined. The researchers now want to use this knowledge to further improve their understanding of the cellular mechanisms of Alzheimer's and, thus, to support the development of strategies for treatment at early stages of the disease.
Benedikt Zott, Manuel M. Simon, Wei Hong, Felix Unger, Hsing-Jung Chen-Engerer, Matthew P. Frosch, Bert Sakmann, Dominic M. Walsh, Arthur Konnerth, A vicious cycle of beta-amyloid-dependent neuronal hyperactivation, Science, August 9, 2019; DOI: 10.1126/science.aay0198
Professor Arthur Konnerth is a member of the cluster of excellence “SyNergy – Munich Cluster for Systems Neurology”, which studies how neurological diseases such as Alzheimer's disease and multiple sclerosis develop. Because the nervous system is extremely complex, the development of neurodegenerative diseases is influenced by numerous processes. A new interdisciplinary approach, system neurology, lies at the heart of the Munich research. The cluster has been funded by the Excellence Initiative since 2012. In addition to the joint applicants TUM and LMU, the German Center for Neurodegenerative Diseases, Helmholtz Zentrum München and the Max Planck Institutes of Biochemistry, Neurobiology and Psychiatry are also involved in SyNergy.
Technical University of Munich
Corporate Communications Center
- Dr. Vera Siegler
- vera.siegler@tum.de
- +49 (0)89 289 23325
- presse@tum.de
- Teamwebsite
Contacts to this article:
Prof. Arthur Konnerth
Hertie Senior Professor of Neurosciences
Institute of Neuroscience of the Technical University of Munich
Tel.: +49-89-4140-3350
Email: arthur.konnerth@tum.de