Size of fat cells can cause metabolic disorders
Why are overweight people more susceptible to illness?
![[Translate to English:] Prof. Hans Hauner Hans Hauner, Professor für Ernährungsmedizin](/fileadmin/_processed_/a/7/csm_Prof_Hauner-8993_725607afff.jpg)
Obesity can be caused by many things: by a genetic predisposition, an unhealthy diet, lack of exercise, a slow metabolism, mental health conditions or the use of certain medications. People with obesity often not only encounter stigmatization by society; they are also likely to suffer from reduced quality of life and a greater risk of secondary diseases. They face a higher incidence, for example, of type 2 diabetes, cardiovascular diseases and various cancers.
But why do people have such an increased tendency to illness when they put on weight? Dr. Julius Honecker and Professor Hans Hauner from the Chair of Nutritional Medicine took a closer look at this question. They were supported in this by the Research Group “Experimental Magnetic Resonance Imaging” led by Professor Dimitrios Karampinos from the Department of Diagnostic and Interventional Radiology of the TUM University Hospital rechts der Isar, and by the lab of Professor Melina Claussnitzer, an institute member at the Broad Institute of MIT and Harvard and assistant professor at Massachusetts General Hospital and Harvard Medical School.
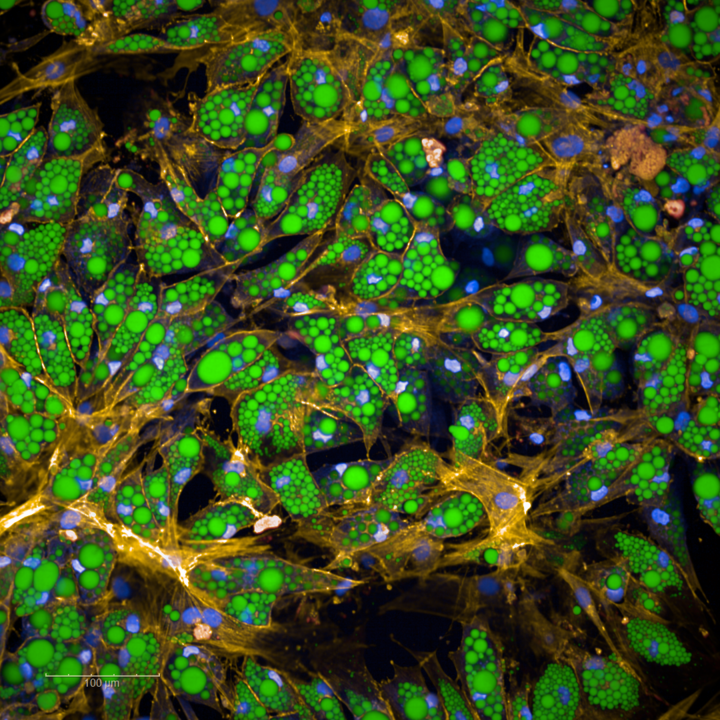
Although the connection between being overweight and being more susceptible to illness has long since been recognized, so far little is known about the importance of fat cell size. The team was now able to show that gene expression – to what extent which genetic information is being used - in adipose tissue is associated with fat cell size and that changes in gene expression in enlarged fat cells are likely to be responsible for the development of metabolic diseases.
“Large fat cells have a significantly altered metabolism and promote among other things the development of type 2 diabetes and cardiovascular diseases,” says Professor Hauner. “This is important evidence that the function of fat cells varies considerably depending on their size.”
The cells shift from energy-consuming to energy-storing
To establish how gene expression depends on the size of the fat cells, the team grouped the adipocytes - large, round fat cells - according to their size and sequenced their RNA. The result was clear: "Adipocyte hypertrophy manifests itself in the altered expression of genes involved in mitochondrial function and fatty acid metabolism," explains Dr. Honecker. Detailed analyses show that the transcriptome – the set of all RNA molecules in a cell – changed from energy-consuming to energy-storing and inflammatory in nature. The cells of very overweight people have an increased tendency to store energy and promote inflammation, which fosters the illnesses already mentioned.
Improved diagnostics without the need for surgery
Looking for ways to examine the fat cell size of patients more easily and without biopsy, Dr. Stefan Ruschke and Professor Karampinos developed and refined a non-invasive magnetic resonance spectroscopy (MRS) method. This also enabled a robust analysis of the fatty acid composition in the adipose tissue and for the first time the concept of a virtual adipose tissue biopsy was established, without the need for a minor procedure to remove the tissue.
With these improved diagnostic methods, used to determine the morphology and composition of the fat tissue, metabolic diseases could in future be diagnosed better and earlier. And it could be done virtually, without any tissue removal. “In this study, it was important for us to establish better methods for simultaneously measuring fat cell size and fatty acid composition in the body,” says Professor Karampinos. “This opens up completely new perspectives, enabling us in future to detect the risks that lie hidden in fat tissue quickly and safely.”
Julius Honecker, Stefan Ruschke, Claudine Seeliger, Samantha Laber, Sophie Strobel, Priska Proll, Christoffer Nellaker, Cecilia M. Lindgren, Ulrich Kulozik, Josef Ecker, Dimitrios C. Karampinos, Melina Claussnitzer, and Hans Hauner (2022): Transcriptome and fatty-acid signatures of adipocyte hypertrophy and its non-invasive MR-based characterization in human adipose tissue. EBioMedicine. DOI: doi.org/10.1016/j.ebiom.2022.104020
The work was supported by the Else Kröner-Fresenius-Stiftung (EKFS), the Kompetenznetz Adipositas and the European Research Council.
Contacts to this article:
Prof. Dr. Hans Hauner
Technical University of Munich
Else Kröner-Fresenius-Zentrum für Ernährungsmedizin
Chair of Nutritional Medicine
Phone: +49 289 249 11
hans.hauner@tum.de


