New link between diet, intestinal stem cells and disease discovered
How unhealthy diet makes you sick

The intestine is essential for maintaining our energy balance and is a master at reacting quickly to changes in nutrition and nutrient balance. It manages to do this with the help of intestinal cells that among other things are specialized in the absorption of food components or the secretion of hormones.
In adult humans, the intestinal cells regenerate every five to seven days. The ability to constantly renew and develop all types of intestinal cells from intestinal stem cells is crucial for the natural adaptability of the digestive system. However, a long-term diet high in sugar and fat disrupts this adaptation and can contribute to the development of obesity, type 2 diabetes and gastrointestinal cancer.
Important role of the intestinal stem cells
The molecular mechanisms behind this maladaptation are part of the research field of the group of Heiko Lickert, professor for diabetes research and ß-cell biology at the Technical University of Munich and head of the Institute for Diabetes and Regeneration Research at Helmholtz Munich.
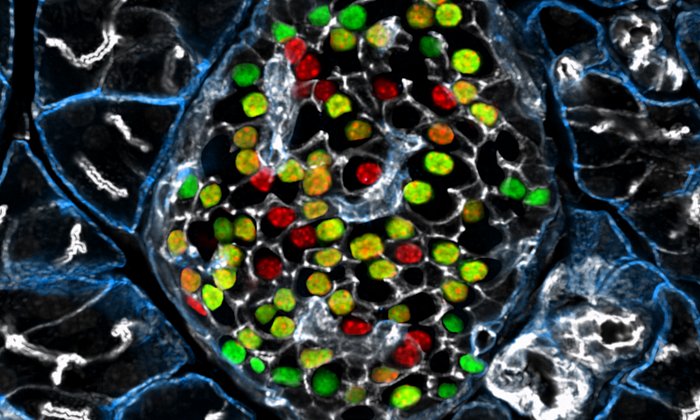
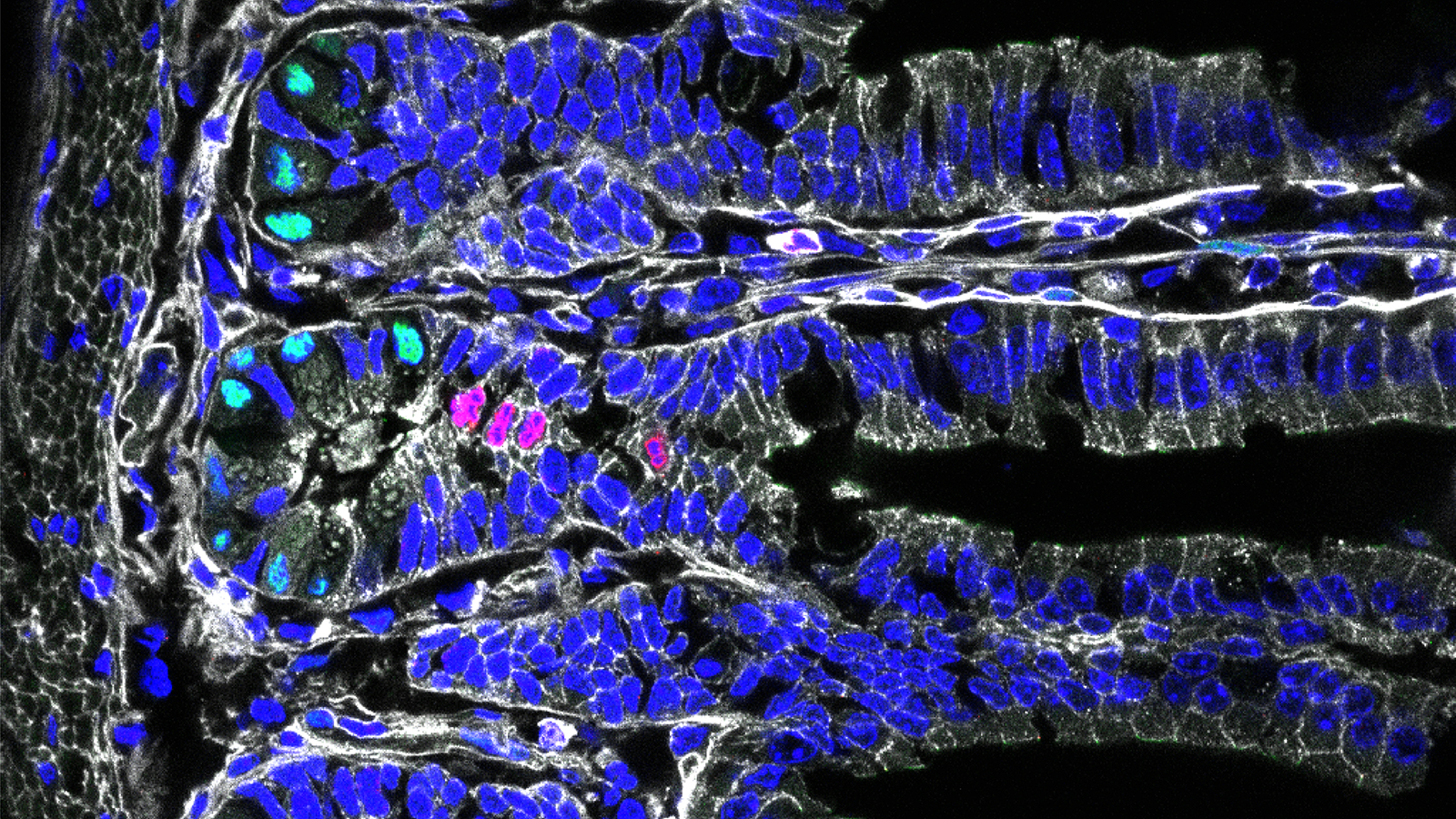
The scientists assume that intestinal stem cells play a special role in the maladaptation. Using a mouse model, the researchers investigated the effects of a high-sugar and high-fat diet and compared it with a control group.
From high-calorie diet to increased risk of gastrointestinal cancer
“The first thing we noticed was that the small intestine increases greatly in size on the high-calorie diet,” says study leader Anika Böttcher. “Together with Fabian Theis’ team of computational biologists, we then profiled 27,000 intestinal cells from control diet and high fat/high sugar diet fed mice. Using new machine learning techniques, we thus found that intestinal stem cells divide and differentiate significantly faster in the mice on an unhealthy diet.”
The researchers hypothesize that this is due to an upregulation of the relevant signaling pathways, which is associated with an acceleration of tumour growth in many cancers. “This could be an important link: Diet influences metabolic signaling, which leads to excessive growth of intestinal stem cells and ultimately to an increased risk of gastrointestinal cancer,” says Böttcher.
With the help of this high-resolution technique, the researchers have also been able to study rare cell types in the intestine, for example hormone-secreting cells. Among their findings, they were able to show that an unhealthy diet leads to a reduction in serotonin-producing cells in the intestine. This can result in intestinal inertia (typical of diabetes mellitus) or increased appetite. Furthermore, the study showed that the absorbing cells adapt to the high-fat diet and their functionality increases, thus directly promoting weight gain.
Important basic research for non-invasive therapies
These and other findings from the study lead to a new understanding of disease mechanisms associated with a high-calorie diet. “What we have found out is of crucial importance for developing alternative non-invasive therapies,” says study leader Heiko Lickert, in summarizing the results.
To date, there is no pharmacological approach to prevent, stop or reverse obesity and diabetes. Only bariatric surgery causes permanent weight loss and can even lead to remission of diabetes. However, these surgeries are invasive, non-reversible and costly to the healthcare system.
Novel non-invasive therapies could happen, for example, at the hormonal level through targeted regulation of serotonin levels. The research group will examine this and other approaches in subsequent studies.
Alexandra Aliluev, Sophie Tritschler, Michael Sterr, Lena Oppenländer, Julia Hinterdobler, Tobias Greisle, Martin Irmler, Johannes Beckers, Na Sun, Axel Walch, Kerstin Stemmer, Alida Kindt, Jan Krumsiek, Matthias H. Tschöp, Malte D. Luecken, Fabian J. Theis, Heiko Lickert and Anika Böttcher
Diet-induced alteration of intestinal stem cell function underlies obesity and prediabetes in mice
Nature Metabolism vol. 3, pages 1202–1216 (2021) – DOI: 10.1038/s42255-021-00458-9
The research was funded by the Helmholtz Association, the German Research Foundation, the German Center for Diabetes Research, the Alexander von Humboldt Society and the European Research Council.
Technical University of Munich
Corporate Communications Center
- Verena Schulz (HZM) / Andreas Battenberg (TUM)
- battenberg@zv.tum.de
- presse@tum.de
- Teamwebsite
Contacts to this article:
Prof. Dr. Heiko Lickert
Technical University of Munich
Professorship of Diabetes and Regeneration Research / Beta-Cell Biology and
Director of the Institute of Diabetes and Regeneration Research at the
Helmholtz Diabetes Center München
Tel.: + 49 89 3187 3760 – heiko.lickert@helmholtz-muenchen.de
Prof. Dr. Dr. Fabian J. Theis
Technical University of Munich
Professorship of Mathematical Modelling of Biological Systems and
Director of the Institute of Computational Biology at Helmholtz Munich
Tel.: +49 89 3187 2211 – theis@mytum.de